The Mayo Clinic is notifying 17,000 heart-surgery patients across the country about a rare but serious infection risk related to a common surgical device (called a heater-cooler) used in open-chest operations. The hospital—which has major campuses in Arizona, Florida, and Minnesota—has identified one such infection in a heart-surgery patient at its St. Mary's Hospital in Rochester, Minn., and has since found many of its heater-coolers contaminated with the microbe.
Consumer Reports learned of the Mayo Clinic's actions, which have not been previously reported, when a patient shared a letter from the hospital with us.
The Food and Drug Administration says that 16 U.S. hospitals, across 10 states, have reported similar heater-cooler incidents. But the agency has not named those hospitals or said which states they're located in, and just a handful have publicly acknowledged the problem.
The Mayo Clinic's letter tells patients that they should contact their primary care doctor if they have questions or experience any symptoms linked to the infection, including night sweats, substantial weight loss over several months, and a persistent, unexplained fever.
The hospital's decision to send that letter to every patient exposed to its heater-coolers was made following guidelines issued in October 2016 by the Food and Drug Administration and the Centers for Disease Control and Prevention for addressing the problem, which first emerged in 2015.
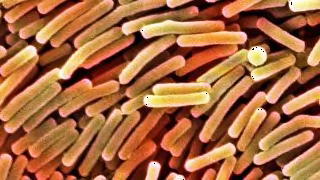
Heater-coolers help regulate a patient's body temperature during surgery. But owing to a design flaw, the machines can spray deadly bacteria—most often a bacteria called non-tuberculosis mycobacteria (NTM), but also other bacteria that grow in water—through their exhaust fans and into patients' open chest cavities during surgery.
That risk is very rare. But the machines are ubiquitous, the confirmed cases are mounting, and the CDC recently linked several separate NTM outbreaks to the same source of contamination: a manufacturing facility owned by the European company LivaNova, which produces 60 percent of all heater-coolers used in the U.S.
Like previous advisories, the October guidelines call on hospitals to take steps to reduce the risk of infection from the devices. Unlike those earlier alerts, the new ones also advise hospitals that use the LivaNova brand heater-coolers to test their machines for contamination and to develop protocols for alerting patients if a device is found to be contaminated or if an outbreak occurs.
What Heart Surgery Patients Need to Know
- If you are undergoing open chest surgery, ask your physician whether a heater-cooler device, or HCD, will be used for the operations, and if so, whether it has been tested for contamination or implicated in any outbreaks.
- If you've already had heart surgery, you and your primary-care doctor should be on the lookout for signs of infection, including irritation around the surgical incision, and unexplained fevers, night sweats, muscle aches, or weight loss, which can emerge as long as four years after surgery.
Alerting Patients
The Mayo Clinic has been using the LivaNova devices since 2012. The hospital convened an emergency meeting at 6:30 a.m. the day after the FDA advisory. Ultimately doctors there decided that, based on the one confirmed case, every patient who had ever been exposed to a heater-cooler at any Mayo hospital should be notified of the risk.
"We just felt strongly that every patient that had had cardiac surgery that could have been exposed to this bacteria should know about it," says Brad Narr, M.D., Mayo's chief of anesthesiology and chair of its Surgical and Procedural Committee.
That decision stands out because experts have been divided over when—or even whether—to warn patients about the problem. The machines are used in hundreds of thousands of operations each year, and some doctors argue that notifying all patients would be overly expensive and could create needless panic.
But others point out that unless patients are alerted, they won't know what symptoms to watch out for—and that lack of awareness could delay treatment and cause worse, potentially devastating, outcomes.
At a meeting in June, doctors from both sides of the issue debated what number of confirmed NTM infections should count as an "outbreak," and thus trigger a response from health officials. Some said one infection, and others said two.
"The Mayo Clinic's response is in line with what we'd like to see all hospitals do," says Lisa McGiffert, director of Consumer Reports' Safe Patient Project. "Health officials have not acted quickly or aggressively enough on this issue, and as a result, too many patients are still being exposed to risk without their knowledge or consent."
Heater-Cooler Alternatives
Hospitals everywhere are struggling to address the heater-cooler concern. Some have taken extreme measures to remove the devices from their operating rooms by drilling holes in the walls and creating separate containment areas that can be connected to the larger surgical theater through a hose.
For now, the Mayo Clinic has resorted to using older, more primitive heater-cooler devices—but only to replace those LivaNova machines that test positive for contamination. The older devices are messier and less efficient, Narr says, in part because they require the use of additional non-sterile equipment, including ice machines, which can introduce additional contamination risks. What's more, the Mayo Clinic had just six of them in storage.
System-wide, Narr says that Mayo has about 23 or 24 LivaNova heater-coolers, which it uses not only for open-chest surgery but also for some patients on life support in its intensive care units. "We can't take all the [LivaNova devices] out of practice immediately, because there's nothing on the market that can respond to our volume," Narr says.
Eventually, the hospital does plan to replace all of its LivaNova heater-coolers with a different brand. But as Narr acknowledged, even that move may not resolve the problem. All modern heater-coolers have the same basic design, and thus are vulnerable to the same device contamination.
"That's absolutely something we've discussed," Narr says. "Is it a problem with this device or with this technology? And are we deluding ourselves in thinking that if we spend this amount of money going forward we can fix it?" The answer, he says, remains to be seen.