
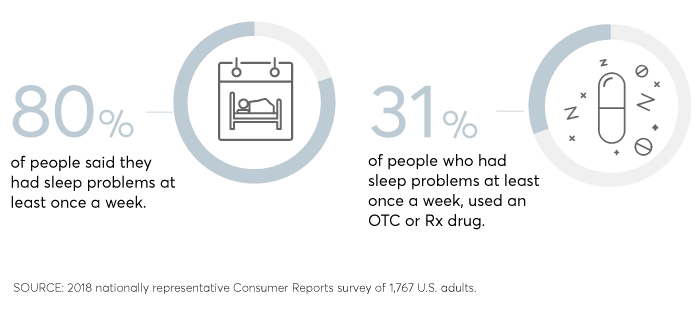
Sleeplessness is complicated—but that hasn't stopped millions of Americans from craving a simple chemical solution.
In a July 2018 Consumer Reports survey of 1,767 U.S. adults, nearly one-third of people who complained of sleep problems at least once per week said they had used an over-the-counter or prescription sleep drug in the previous year.
But many of those people don't get the restful night's sleep they seek. Only about a third of people who took sleep medications—either OTC or prescription—reported very good or excellent sleep on those nights.
And they often don't wake refreshed: Nearly 6 out of 10 people taking sleep medications reported side effects such as feeling drowsy, confused, or forgetful the next day. Three percent admitted to dozing off while driving.
Among the most common reasons Americans gave for taking a sleep drug is that they thought it would work better than other approaches.
But drugs aren't the cure-all many people expect.
"In terms of improvement of total sleep time, the benefits of sleep medications are pretty modest," says Michael Sateia, M.D., emeritus professor of psychiatry at the Geisel School of Medicine at Dartmouth. Plus, some of the most commonly used drugs, notably OTC sleeping pills, should never be used long-term, and have little published research backing up even their short-term use, Sateia says.
And all sleep medications—including OTC products—have important and often underappreciated risks.
What's more, our survey found that about 40 percent of people who take sleep aids use the drugs in potentially harmful ways—by, for example, taking them more often or longer than recommended, or combining them with other medications or alcohol.
Here's what you need to know about the benefits and risks of the drugs, the situations when taking them makes the most sense, and how to use them safely.

How Sleep Drugs Work
Different types of sleep medications affect the brain and body in different ways.
All OTC sleep drugs rely on older antihistamines—diphenhydramine (Sominex, ZzzQuil, and generic) or doxylamine (Unisom SleepTabs and generic)—which have drowsiness as a side effect.
A wider variety of sleep medications are available by prescription.
The oldest type is anti-anxiety drugs, such as temazepam (Restoril and generic) and triazolam (Halcion and generic), from a class of medications called benzodiazepines, or "benzos." These drugs make you feel drowsy or sedated by slowing activity of the brain and central nervous system.
The next generation of medications, the so-called Z drugs, including eszopiclone (Lunesta and generic), zaleplon (Sonata and generic), and zolpidem (Ambien, Edluar, Intermezzo, Zolpimist, and generic), target the same brain receptors. But they do so more selectively, which is thought to reduce some of the side effects, such as lingering grogginess and the risk of becoming dependent.
The newest type of prescription sleep drugs, ramelteon (Rozerem) and suvorexant (Belsomra), affects brain chemicals that regulate the sleep-wake cycle.
Doctors also sometimes treat sleeplessness with an older type of antidepressant—most commonly trazodone—because it also has sleepiness as a side effect. Although the Food and Drug Administration has not approved trazodone for insomnia, doctors can legally prescribe a drug for any reason they see fit.
See "A Quick Guide to Sleeping Pills," below, for more information on different sleep drugs.
Limited Benefits
One thing all these medications have in common: limited benefits.
Sleep drugs can help people fall asleep faster or return to sleep if they wake up in the middle of the night, but the benefits are usually modest, says Daniel Buysse, M.D., UPMC professor of sleep medicine and professor of psychiatry at the University of Pittsburgh School of Medicine. "Most only increase total sleep time by about 20 to 30 minutes," he says.
Buysse, along with Sateia and other researchers, reviewed 46 studies of sleep drugs for the most recent treatment guidelines from the American Academy of Sleep Medicine (AASM) (PDF), published in 2017.
Some drugs have even less benefit. For example, studies show that, on average, people taking ramelteon fall asleep 9 minutes faster than those taking a placebo; those taking suvorexant got only 10 minutes more shut-eye than those who took a placebo.
Most surprising: Some commonly used sleep drugs haven't been shown to help at all.
Take OTC products, for example. About 1 out of 5 people from Consumer Reports' nationally representative sample—which translates to about 50 million U.S. adults—reported using these in the past year. But these old-school antihistamines simply haven't been studied for sleep.
"People think that because something is available over the counter it is safe and effective," Buysse says. "But there's shockingly little evidence on the efficacy of these drugs."
Similarly, although doctors commonly prescribe the antidepressant trazodone for sleeplessness, there's little data to support that use.
Because trazodone and OTC drugs such as diphenhydramine have known risks and there's little evidence of their benefit when used for sleep, the AASM treatment guidelines don't recommend either type of medication for chronic insomnia.
The 'Morning After' Effect
As shown in "A Quick Guide to Sleeping Pills," below, all sleep medications have side effects—most commonly causing people to feel drowsy, dizzy, mentally less sharp, or unsteady the next day. More rarely, people taking sleep medications have reported sleepwalking, hallucinations, memory lapses, and odd behavior, such as eating or driving while asleep. With most medications, long-term use can be habit-forming and stopping the drugs can lead to a rebound effect, where insomnia actually gets worse.
In the Consumer Reports survey, one of the most common reasons people gave for taking OTC drugs is that they thought they were safe. But the sedating effects of OTC drugs frequently linger into the next day. In fact, 40 percent of respondents taking an OTC sleep drug said that they felt foggy or drowsy the next morning, compared with 32 percent of those taking a prescription drug. In addition, the older antihistamines used in OTC sleep drugs can cause confusion, constipation, dry mouth, and trouble urinating.


The side effects of sleep drugs—particularly excessive drowsiness, unsteadiness, and confusion—tend to worsen as people age. "Older people metabolize these drugs more slowly and so are at increased risk of impairment," Sateia says. That's particularly concerning when you consider that nearly 1 out of 10 respondents to the Consumer Reports survey who took sleep aids reported feeling uncoordinated, tripping, or falling.
Indeed, research shows that older people who take sleep drugs are more likely to fall and suffer broken bones and brain injuries. For example, a 2017 analysis of 4,669 people 65 and older found that, over a two-year period, people taking sleep drugs recommended by their doctor were 34 percent more likely to fall than those who weren't.
Women may also be more susceptible to next-day drowsiness because they clear some drugs from their bodies more slowly than men. To address that difference, a few years ago the FDA required drugmakers to cut in half the recommended dosages of medications containing the Z drug zolpidem (Ambien, Ambien CR, and generic; Edluar; Intermezzo; and ZolpiMist). The agency said that physicians should also consider prescribing lower doses for men. (Drug labels already called for lower doses in older people.)
But many doctors ignore these recommendations, according to a study in the September 2018 issue of JAMA Internal Medicine. Researchers analyzed data on adults taking zolpidem from the U.S. Medical Expenditure Panel Survey, the largest publicly available national survey of prescription drug use. They discovered that about two-thirds of women and people 65 and older were taking higher doses of the drug.
The Risky Business of Sleep Drugs
The risks of sleeping drugs are magnified when the medications are misused. For example, combining sleeping pills with other sedatives, prescription pain drugs, or even alcohol "puts you at real risk for serious side effects such as dangerously slow breathing and mental and physical impairment that may linger into the next day," Sateia says.
Buysse says that people should take warnings to use sleeping pills only as directed seriously. For example, most sleep drugs caution that you shouldn't take them unless you can spend 7 or 8 hours in bed.
"I especially worry about people who take a sleep medication with 4 hours or less available to them to sleep," he says. "After 4 or 5 hours, you can still have the drug circulating in your blood, so you will be less alert at precisely the time you need to be doing things that require your concentration."
"It's the exact same reason that people shouldn't drink and drive," Buysse says. "Even if you feel relatively alert, you could still be a danger to yourself and others."
Research backs up that concern. People prescribed sleeping pills are nearly twice as likely to be in a car crash, according to 2015 study in the American Journal of Public Health (PDF) that looked at the medical and driving records of nearly 410,000 adults. The researchers estimated that people taking sleep drugs were as likely to be in a car crash as those driving with a blood alcohol level over the legal limit.
Buysse says that he is also concerned by the number of respondents in the Consumer Reports survey who said that they exceeded the recommended dose, combined different types of drugs, or took sleep drugs with alcohol and other recreational drugs.
In our survey, one out of 10 Americans who take prescription sleep drugs reported taking an opioid to sleep, a drug that is especially dangerous when combined with other sedatives or alcohol. All these drugs can cause your breathing to become more slow and shallow, Buysse says, so "if you combine an opioid with a benzodiazepine or alcohol, you could stop breathing altogether."


Safer Use of Sleeping Pills
Because of the limited benefits and important risks of sleeping pills, the drugs should be used with great caution.
In general, sleeping pills should be reserved for short-term insomnia—such as that caused by jet lag, anxiety after the death of a family member, or stress from a job loss—Sateia says.
For sleeplessness lasting more than a couple of weeks that is affecting your daily life, the AASM recommends cognitive behavioral therapy for insomnia (CBT-I), a form of counseling that helps people identify and replace thoughts and behaviors that contribute to sleep problems. "CBT-I is at least as effective as medications, the benefits are long-lasting, and it's manifestly safer than any pill," Sateia says.
Some people with severe sleep problems may benefit from taking a prescribed sleep drug for a few weeks in conjunction with CBT-I and then gradually tapering off the medication as the therapy takes effect.
In the Consumer Reports' survey, 40 percent of Americans who take OTC drugs and 55 percent of those who take prescription drugs have been taking the meds for a year or longer. But long-term use is typically not recommended because the drugs may become less effective over time and, in some cases, people become hooked, or dependent on them. They may crave the drug, take more than recommended, and not be able to stop even if the drug is no longer helping or is causing side effects.
"Very few people need sleep drugs long-term," Sateia says. "And in those cases patients should be monitored carefully to make sure that the drugs are still effective and that they are not developing a dependency."
For the limited situations where a prescription or OTC sleep drug is warranted, follow these precautions:
- Tell your doctor about all the medications you take, including supplements and herbal products (such as St. John's wort). Many common drugs, such as certain antibiotics and antidepressants, can interact dangerously with sleep drugs.
- Take the drugs only if you have 7 or 8 hours of time available to sleep. Even if that much time has passed, don't drive if you feel drowsy.
- Don't take a sleep drug if there's a chance you may need to be up and alert during the night—if your job requires you to be on call, for example, or you are the sole caretaker for young children.
- Do not take an extra dose if you wake up in the middle of the night.
- Never mix sleeping pills with alcohol, recreational drugs, or other sleep drugs or supplements, including OTC nighttime pain relievers and antihistamines, such as diphenhydramine in Benadryl Allergy, that also act as sedatives.
- Start with the lowest recommended dose, especially until you know how the drug affects you.
- Be cautious about long-term use. Taking sleep drugs regularly for longer than two weeks can breed dependence and raise the risk of adverse effects.
Examples: Flurazepam, quazepam (Doral), temazepam (Restoril and generic), and triazolam (Halcion and generic).
Side effects and risks: Next-day drowsiness, dizziness, and feeling uncoordinated or mentally less sharp. Benzos slow breathing and can worsen sleep apnea or other breathing problems. Long-term use can be habit-forming, and insomnia may worsen when stopping the drug. More rarely, people have reported behaviors such as walking, driving, eating, making calls, or having sex while not fully awake.
Recommended for: Helping people to fall asleep faster (triazolam); helping people to fall asleep faster and sleep longer (temazepam). The American Academy of Sleep Medicine (AASM) did not find sufficient evidence to make recommendations for flurazepam and quazepam.
Precautions: Be cautious driving the day after taking a benzo until you know how it will affect you. Don't take the drugs with opioids or alcohol because that increases the risk of slow or difficult breathing and death.
Examples: Eszopiclone (Lunesta and generic), zaleplon (Sonata and generic), and zolpidem (Ambien and generic). Zolpidem is also available as a dissolvable tablet (Edluar and Intermezzo) and an oral spray (ZolpiMist).
Side effects and risks: The same as for benzos, although research suggests that side effects of Z drugs are generally milder and less common. Higher doses increase the risk of next-day impairment.
Recommended for: Helping people to fall asleep faster (zaleplon); falling asleep faster and staying asleep longer (eszopiclone and zolpidem).
Precautions: To reduce the risk of side effects, start at a low dose. The extended-release version of zolpidem, Ambien CR, carries a warning not to drive the next morning. Don't combine with alcohol, opioids, or other sedatives.

Examples: Ramelteon (Rozerem) and suvorexant (Belsomra).
Side effects and risks: Ramelteon can cause dizziness, nausea, fatigue, and headache. The most common side effects of suvorexant are headache, dizziness, and lingering sleepiness. The drug may also cause sleepwalking or other nighttime behaviors, sleep paralysis, hallucinations, and worsening of depression and suicidal thoughts.
Recommended for: Falling asleep faster (ramelteon); staying asleep longer (suvorexant).
Precautions: People taking suvorexant may be less alert the next day, and the risk of impairment is greater with higher doses. The 20-mg dose of suvorexant carries a warning not to drive the next day.
Examples: Doxepin (Silenor and generic) and trazodone.
Side effects and risks: Both drugs may cause nausea and dizziness. In addition, trazodone has a long list of other potential side effects, including next-day drowsiness, abnormally low blood pressure, heart-rhythm disorders, a persistent erection in men, and suicidal thoughts and behaviors in children and adolescents.
Recommended for: Staying asleep longer (doxepin). Because of a lack of evidence that trazodone is effective for insomnia and uncertainty about the risks, the AASM does not recommend the drug for treating insomnia.
Precautions: People 55 and older should be cautious taking trazodone because they are more susceptible to abnormal heart rhythms and falls caused by dizziness or drowsiness. Use the drug cautiously if you have heart, liver, or kidney disease, or weakened immunity, and avoid it if you've had a heart attack.

Examples: Diphenhydramine (Sominex, ZzzQuil, and generic) and doxylamine (Unisom SleepTabs and generic).
Side effects and risks: Next-day drowsiness, constipation, dizziness, difficulty urinating, vision problems, and feeling mentally less sharp.
Recommended for: Due to limited evidence of benefits and known risks, the AASM does not recommend diphenhydramine for insomnia. The experts Consumer Reports consulted with don't recommend doxylamine, for the same reason.
Precautions: Like prescription sedatives, OTC sleep drugs cause next-day drowsiness and slowed breathing, so follow the same precautions you would with those medications. To avoid inadvertently taking too high a dose, don't combine OTC sleep drugs with other medications that contain older antihistamines, including allergy drugs such as Benadryl; nighttime pain relievers such as Advil PM and Tylenol PM; and cough-and-cold drugs such as Robitussin Severe Multi-Symptom Cough Cold + Flu Nighttime and Sudafed PE Severe Cold.
How to Get a Good Night's Sleep
Not getting enough Zzzzs? 'Consumer 101' TV show host, Jack Rico, gets expert CR tips on how to fall asleep faster and wake up more rested.
