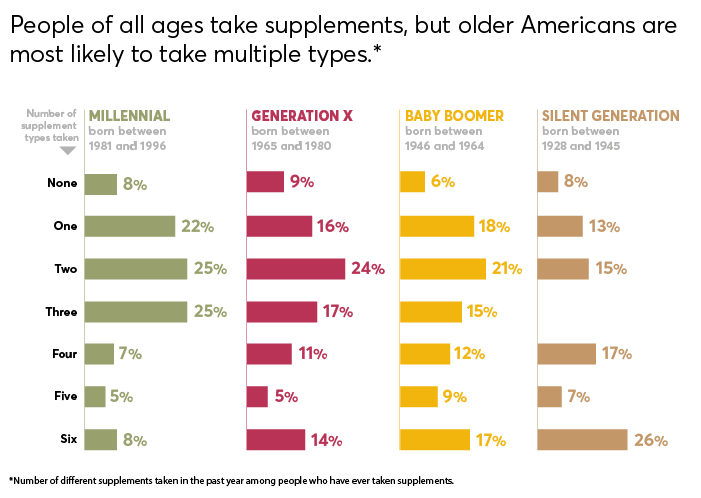
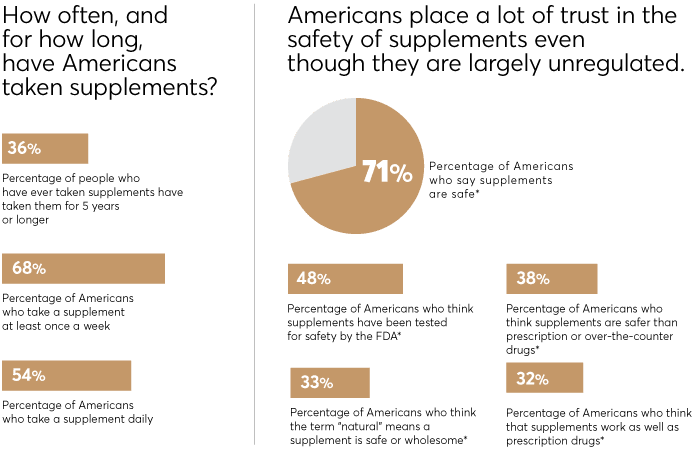
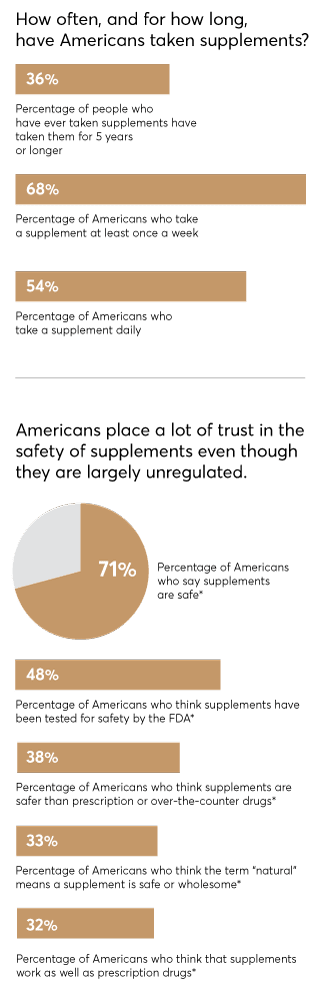
The promise of supplements is pretty powerful: That they'll ease your pain, boost your immunity, settle your stomach, strengthen your heart, sharpen your memory, and more. Little wonder, then, that supplement sales in the U.S. reached $46 billion in 2018 and are predicted to exceed $52 billion by 2020. In fact, 68 percent of Americans take supplements at least once a week, according to a recent nationally representative Consumer Reports survey of 2,006 adults.
So it's perhaps remarkable that there's not, to date, a lot of solid evidence that supplements do what people hope they will. Manufacturers are not required to demonstrate to the government that their products are effective or safe before they are sold—as they must with prescription and over-the-counter drugs, such as Advil.
People have long used botanicals like echinacea to help with infections such as the common cold, and turmeric to curb inflammation and pain. But both—despite showing promising results in lab settings—fail to provide significant benefits over a placebo in most structured clinical trials. For example, an August 2019 review looked at 19 clinical trials of turmeric and its active compound, curcumin, to see whether either could reduce inflammation in patients with chronic inflammatory diseases, such as osteoarthritis. It found no sign that either had a significant effect.
"There are simply not good studies that support a lot of supplement claims," says Robert McLean, M.D., a rheumatologist at Yale New Haven Hospital and president of the American College of Physicians.
Among the most widely used supplements in our survey are multivitamins and vitamins, followed by fish oil, calcium, and probiotics. But the market for botanicals—a category of supplements derived from plants—has grown from about $4.2 billion in 2000 to more than $8.8 billion in 2018, according to Nutrition Business Journal. Our survey found that among people who had taken supplements in the previous year, 38 percent had taken at least one botanical.
One reason supplement sales may remain stubbornly robust is because "there's a strong placebo effect," McLean says. "People will feel better if they think they're going to feel better."
That means consumers are often left guessing about efficacy. Suyash Raj, a research technician, concedes "it's very hard to quantify" the exact effect of the two botanical supplements he takes. But he has looked at studies to research their safety, and says they seem to help him with stress. "I feel better," he says— adding that "of course that could be a placebo [effect]." Similarly, Stacy Bond, a writer and public radio producer, now takes multiple supplements to help deal with a few health concerns. While she says she's not sure they're all effective, "there's something, I think, maybe before I was lacking that I'm now getting from the supplements."
Also muddying the evidentiary waters: Research shows that people who buy supplements tend to be healthier than the average person in the first place.
CR's survey also found that 48 percent of Americans believe supplements have been tested for safety by the Food and Drug Administration, even though the agency does not do comprehensive testing of them. An additional 71 percent believe supplements are safe. But many pose dangers, says Pieter Cohen, M.D., an internist at Harvard Medical School who has studied supplements extensively. One 2015 study by government researchers found that supplement use led to more than 23,000 emergency room visits a year, most often because of heart problems triggered by supplements taken for weight loss and energy.
"There's a lack of oversight, a lack of safety, and a lack of rigorous science," says Daniel Lasoff, M.D., a medical toxicologist in the department of emergency medicine at the University of California, San Diego.
CR tested turmeric and echinacea, and our findings revealed problems with potency and purity even among some of the most widely used brands. (See "CR Tests: Turmeric and Echinacea.")
And yet "dietary supplements have benefits to offer," says Richard van Breemen, Ph.D., a professor of pharmaceutical sciences and the director of the Linus Pauling Institute at Oregon State University, which focuses on vitamin, mineral, and plant-derived chemical research. He cites black cohosh (see "A Guide to 10 Popular Botanicals") as one example where some women say it eases their menopausal symptoms, even though scientists don't understand how it works. But before supplements can be used effectively to address specific ailments, "we need more research on botanicals to figure out the optimum preparations and dosages," he says, adding that more information is also needed on their long-term safety.
The Potency of Plants
The top reasons for taking botanicals, aside from turmeric and echinacea (see "Lifestyle Changes That Work—Without the Risks"), were "to improve or maintain overall health" and "to strengthen general immunity," according to our survey.
Plants have, in fact, been used for medicinal purposes for thousands of years. Today, approximately half of prescription medicines in use are derived from plants, microbes, and fungi found in the natural world, according to van Breemen at Oregon State. But the key difference is that prescription drugs used to treat cancer and other diseases are standardized, with each dose containing the exact same quantity of active ingredients.
Such standardization is not mandatory for supplements in the U.S. This is partly why figuring out which botanicals are safe and effective—and which may be useless or risky—can be particularly challenging. Botanicals include turmeric, St. John's wort, green tea extract, and valerian. (CBD, or cannabidiol, is botanical in origin, which is why many people think of it as a botanical supplement. But the FDA currently considers it illegal to sell the trendy compound from the cannabis plant as a supplement because it's approved as a drug to treat rare forms of epilepsy. Get more information at our Cannabis & CBD Guide.)
Part of the challenge is that plant-derived pills can be extremely potent—and that potency can be dangerous. According to Harvard's Cohen, some botanicals—red yeast rice, used for heart problems, and the now-banned ephedra, once found in weight-loss supplements—can deliver the same amount of a "natural" chemical as a prescription drug. That means that they can have side effects that are similar to (though less predictable than) those of drugs, according to Cynthia Rider, Ph.D., a toxicologist at the National Toxicology Program at the National Institute of Environmental Health Sciences (NIEHS).
Take, for example, vinpocetine, a synthetic substance derived from a chemical found in the Vinca minor plant. In some countries it's sold as a prescription drug to treat stroke or cognitive impairment. However, in the U.S. it isn't an FDA-approved prescription drug but is sold as a botanical ingredient in some memory supplements. Vinpocetine can cause dangerously low blood pressure, and birth defects or miscarriage if taken by a pregnant woman.
The form and source of raw ingredients also vary: Research indicates that some overseas suppliers of turmeric spice have sometimes intentionally added lead to it to brighten its distinctive color, making it especially risky when consumed regularly. The same plant grown in California and India can have different levels of active compounds. Additionally, many plants can suck up heavy metals from the soil as they grow, turning a safe plant dangerous. And botanical supplements are sometimes manufactured in unsanitary conditions, allowing bacteria to be introduced into the products.
One particularly harmful side effect linked to supplements is liver damage. In 2004, the percentage of all liver damage cases in the U.S. involving herbal or dietary supplements was estimated at 7 percent; by 2014, that number was estimated to have climbed to 20 percent, mostly because of green tea extract in weight loss supplements, and steroids illegally added to bodybuilding supplements.
Active compounds in supplements can also amplify the effects of prescription drugs or render other medications ineffective. Ginkgo biloba, for example, can thin the blood, increasing the effect of a prescription blood thinner, such as warfarin.
"I have totally seen this perception of ‘natural' equals safe," says Rider at the NIEHS. "I think it is not a fair assumption."
The results of CR's tests of echinacea and turmeric conform with the kinds of problems also seen by Tod Cooperman, M.D., the president and founder of ConsumerLab.com, an organization that regularly tests supplements and certifies products that pass its quality control tests.
Representatives of the supplement industry point to products with major problems or inconsistencies as outliers. "There are some supplements in the marketplace that either don't contain what their label says they are supposed to contain or they contain things that are not supposed to be there, but that is a very small minority of the industry," says Steve Mister, president and CEO of the Council for Responsible Nutrition, a dietary supplements trade organization.
Yet according to Cooperman, more than 25 percent of the botanical supplements that ConsumerLab.com examines fail the organization's testing, either because of bacterial or heavy metal contamination or because they don't contain what's listed on the label. Botanical supplements are much more likely to fail these tests than mineral or vitamin supplements because, he says, plants often have different levels of active compounds and can pick up contamination from the environment where they were grown or packaged.
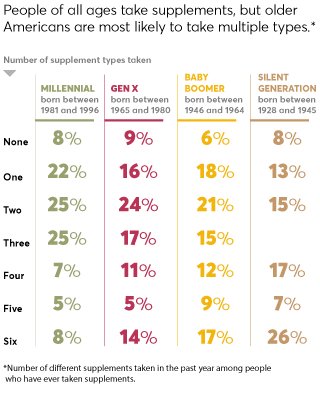
Protections on the Way?
The FDA doesn't test supplements for safety or screen them for contaminants before they are sold, but it does require that supplements contain the ingredients listed on their labels. However, the agency doesn't perform comprehensive tests to verify compliance. And although complying with U.S. Pharmacopeia (USP) quality standards—including tests for identity, potency, and purity—is mandatory for prescription and over-the-counter drugs, doing so is voluntary for supplements. Most manufacturers do not opt in—especially when it comes to botanicals, where standardization can be a challenge, proprietary blends are common, and there is less agreement about appropriate dosage.
The FDA generally requests a recall of a supplement if it receives a large number of reports (from consumers, doctors, manufacturers, and others) indicating that it might be causing serious illnesses—because of contamination with salmonella or prescription-drug ingredients, for example—or if FDA tests indicate a problem. The agency has posted about 34 such recalls for dietary supplements over the past two years, according to an FDA database. But a product that simply doesn't contain what it should could potentially fly under the radar indefinitely, Harvard's Cohen says.
Earlier this year, the FDA indicated that it would be taking steps to more strictly regulate the supplement marketplace, and launched a new online list to warn consumers about certain supplement ingredients. An FDA spokesperson also told CR that the agency has continued to step up enforcement against bad actors. It cited, among other examples, the recent seizure of 300,000 containers of dietary supplements manufactured in substandard conditions, and recalls of drugs illegally sold as supplements.
Even though these increased efforts to enforce the law are a positive step, consumers have a right to expect the FDA to have been doing this all along, says Chuck Bell, who has led CR's advocacy work on supplements for 20 years. Nor does it change the fact that—over several decades—the agency has been slow to push for changes to regulations that experts say continue to leave too many unsafe products on the market. "Progress has been glacial," Bell says.
What Consumers Can Do
When it comes to safety, the onus still largely falls on consumers to be advocates for themselves. Indeed, industry representatives say consumers should do their own research to find safe products. "You have some responsibility to become informed about the supplements you use," says Michael McGuffin, president of the American Herbal Products Association, an industry group. "We recommend consumers consult with their healthcare practitioner."
Before you try any supplement, Bell says, you should discuss it with your primary care provider and seek out trustworthy information from sources such as MedlinePlus.gov, the NIH's health portal for consumers.
A frank assessment of your own habits is probably in order, too. As much as supplement-takers might believe that supplements are safe and effective, many mainstream healthcare practitioners say the products are a waste of money. For those committed to continuing supplement use, see "How to Choose Products Wisely" to help you spot labels that can identify more trustworthy products.
"One day I envision that this industry will have products that are shown to be both safe and effective," says van Breemen at Oregon State. "But we're not there yet."

What the FDA Does,
and Doesn't, Do
Even though some supplements are as potent as certain prescription drugs, different regulations apply. The Food and Drug Administration doesn't need to approve supplements as effective or safe before they are sold, as it does with drugs.
According to the Dietary Supplement Health and Education Act of 1994—the primary law that governs these products—supplement ingredients that were on the market before 1994 are presumed safe. Companies can assert that a product is safe for use as long as they can provide some evidence that its ingredients were used before 1994 or are currently present in the food supply. While manufacturers are supposed to notify the FDA when they start selling a supplement that contains a new ingredient, they often don't, according to Chuck Bell, in the advocacy division of CR.
The FDA technically requires that supplements contain what they say they do and aren't contaminated with harmful substances. But supplements have exploded in popularity since 1994. Current regulations can't keep consumers safe, Bell says. "We're dealing with a much larger industry, and the number of people that are employed by the FDA to oversee that industry has not kept pace," he says.
Editor's Note: This article also appeared in the December 2019 issue of Consumer Reports.
Consumer Reports has updated this article to clarify the process the FDA uses to request recalls of supplements.





